|
OPN and PPARG staining was performed using protein-specific primary antibodies and HRP-labeled secondaries, and visualized with Vector NovaRed Substrate Kit (Vector Labs) (bar = 100 µm). Masson’s trichrome stains collagen blue (left column) and Oil Red stains intracellular lipid deposits red (third column). Differentiation was further verified using histological and immunohistochemistry staining (D) of matrix evolution by encapsulated hMSCs. Immunoblots (A) were quantified with ImageJ software and CBFA1 (B) and PPARG (C) expression levels over the 21-day culture period were normalized to β-actin expression (PEGDM: X t-butyl functionalized: closed diamonds phosphate-functionalized: open circles), error bars represent one standard deviation. CBFA1, PPARG, and β-actin expression of hMSCs encapsulated in control, t-butyl, and phosphate-functionalized PEG hydrogels and cultured for 0, 4, 10, and 21 days in control media. Strategies using simple chemistry to control complex biological processes would be particularly powerful as they could make production of therapeutic materials simpler, cheaper and more easily controlled.Įncapsulation of hMSCs in phosphate and t-Butyl functionalized PEG hydrogels alters CBFA1 and PPARG expression. To our knowledge, this is the first example where synthetic matrices are shown to control induction of multiple hMSC lineages purely through interactions with small-molecule chemical functional groups tethered to the hydrogel material. Hydrogels were formed using sufficiently low concentrations of tether molecules to maintain constant physical characteristics, encapsulation of hMSCs in three dimensions prevented changes in cell morphology, and hMSCs were shown to differentiate in normal growth media, indicating that the small-molecule functional groups induced differentiation. The work presented here demonstrates that encapsulated human mesenchymal stem cells (hMSCs) can be induced to differentiate down osteogenic and adipogenic pathways by controlling their three-dimensional environment using tethered small-molecule chemical functional groups. In 2006, we started in order to make learning organic chemistry fast and easy.Cell-matrix interactions have critical roles in regeneration, development and disease. 
#fail During this entire time, he always loved helping students, especially if they were struggling with organic chemistry. In between, he did NOT compete at the 1996 Olympics, make the Atlanta Braves opening day roster, or become the head coach of the Indiana Pacers, as he had intended. 
After all of that, he (briefly) worked as a post-doctoral assistant at Syracuse University, working on novel organic light-emitting diodes (OLEDs). Prior to all of this, he was a chemist at Procter and Gamble. His research focus was on novel pain killers which were more potent than morphine but designed to have fewer side effects. Mike Pa got a bachelors degree in chemistry from Binghamton University, a masters degree in organic chemistry from the University of Arizona and a PhD in bio-organic chemistry from the University of Arizona. Some chemists refer to ethers as “dead molecules” because of their low reactivity.Īnd now for some crazy functional groups….ĭr. They reactivity depends on the type of alkyl halide (F, Cl, I, Br), its substitution (primary, secondary, tertiary) and the desired reaction (SN1, SN2, E1, E2).Īlcohols, Amines, and Thiols: these are generally very good nucleophiles, as the heteroatoms have lone pairs which will attack an electrophile.Įthers: do not undergo many organic reactions themselves, but sometimes can be the product of a reaction. Appendix I - Hazards Of Functional Groups Alcohols: Aldehydes: Aliphatic Amines: Aliphatic and Aclicyclic Hydrocarbons: Alkali and Other Reactive Metals. 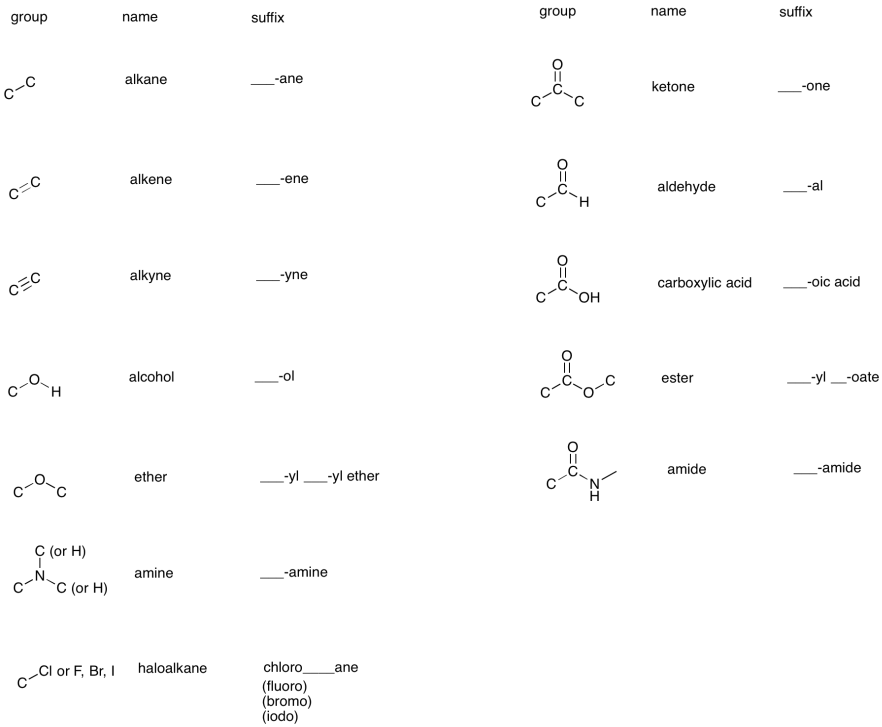
These can participate in nucleophilic substitution reactions and elimination reactions. Generally speaking, if there is an electron withdrawing group attached to the carbonyl carbon, that carbonyl will be more electrophilic and more reactive.Īlkyl Halides: alkanes which are connected to a halogen atom (F, Cl, I, and Br) are good electrophiles. The important point here is to know which types of carbonyls are more electrophilic and which ones are less. While there are different variations which can make the carbonyl more or less reactive, the basic functional group is still the same. It is one of the more important electrophiles you will see in this course. Carbonyls: a “carbon double bond oxygen” is a carbonyl.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
